By: Peter Bennett & Ian Whitehall
Introduction
Changing sample vessel format is an area of Discovery research that can cause problems: sample tracking, sample loss and robotic referencing are among the many drawbacks of having to perform a transfer step.
Genevac set out to attempt to “automate” the transfer step within their evaporation systems and developed several concept designs. The final iteration of this is now launched as the SampleGenie™ system, shown in figure 1.SampleGenie comprises a specially designed large volume glass flask that securely seals to a small storage vial, dramatically increasing its capacity. This allows a purified or other large volume sample to be evaporated directly into the small vial. The whole assembly is loaded into the Genevac HT or EZ-2 evaporation system and using a pre-written method evaporated to dryness. SampleGenie guides the compound into the storage vial directly, thusavoiding the costly transfer step that has traditionally been the cause of problems, and reduces the overall processtime by between 0.5 to 1 day in the typical laboratory. If an amorphous anhydrous powder is required at the end of this step then an HT series evaporator with LyoSpeed™ can be used to concentrate the large volume into the vial, and then Lyophilise the remaining volume to leave a powder in the vial. A concentration only version is also available for users requiring a wet sample.
Figure 1 – SampleGenie
Selection of systems shown with arrangement sketch below.
Key
Dark blue – flask
Red – seal
Light blue – vial
Green – adaptor

SampleGenie technology is applicable in many areas where reformatting is an issue in the first instance.Genevac is applying the SampleGenie solution within:
Medicinal Chemistry
Transfer to the storage vial has traditionally been a wasteful process, hitherto this has either been done by scraping the dried compound into the storage vial or going through a process of resolublising and transferusing a liquid handling workstation, and then a further evaporation step. Both of these are time consuming and have potentially bad yields. Central to both of these approaches has been the Genevac HT and EZ-2 parallel evaporation systems, using precise control these systems dry the compounds well but have not aided in their transfer to the desired storage vial.
Drug Metabolism
When carrying out drug metabolism studies, similar issues may arise in that large volumes of solution (e.g.excreta or extracts) containing a radiolabelled test compound and its metabolites are generated. These mustbe dried and transferred to a small vial before the samples are reconstituted in smaller volumes and analysed e.g. by HPLC with radiochemical detection. The following study was conducted by the Departmentof Drug Metabolism and Pharmacokinetics at Servier Research and Development Ltd who set out toevaluate SampleGenie to determine if it is suitable for their processes. Their aim was to replace several manual steps. Quantitative recovery of the sample is key to their work in evaluating the drugs and metabolites under test. This paper sets out the methods and results from their investigation.
Methods
Two methods were programmed into the Genevac HT-4X. The first was used to evaporate 40ml water to dryness, the second to evaporate 40ml Acetonitrile to dryness. Recovery was assessed for the evaporation of [14C]-X in water and in acetonitrile. The molecule used in this study cannot be named for commercialreasons.
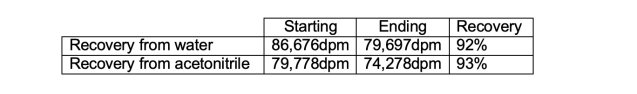
Recovery from Water
40ml of water was spiked with approximately 100,000 dpm [14C]-X. This solution was placed in the upper flask of a SampleGenie holder and the correct Genevac method was run. After 12 hours the sample wasdry. It was then reconstituted into 1ml water. Recovery after reconstitution was assessed.
Recovery from acetonitrile
40ml of acetonitrile was spiked with approximately 100,000 dpm [14C]-X. This solution was placed in the upper flask of a SampleGenie holder and the correct Genevac method was run. After 4 hours the sample was dry and it was thenreconstituted into 1ml methanol. Recovery after reconstitution was assessed.
Results
A final visual experiment was also conducted. 50ml water was spiked with 100µl green food dye (figure 2). This sample was then evaporated to dryness using the ‘water’ Genevac method. All the green dye appears to be trapped in the base of the collection vial, with none apparent on the flask or seal.
Figure 2 – Green food dye in water before concentration (shown left), and then concentrated into the vial (shown right)

Conclusions
In both sets of conditions tested, the SampleGenie system managed to achieve a step yield of 92-93%,this compares very favourably with the anticipated yield from either manual transfer or resolublisation and liquid handling. In addition to the excellent recoveries, another major advantage to using SampleGeniewas the significant reduction in processing time. Although there is no XYZ coordinate programming involved,SampleGenie is liquid handling Automation!
About the Authors
Peter Bennett is a Senior Research Scientist within the Department of Drug Metabolism and Pharmacokinetics, Centre for Biopharmacy Research, Servier Research and Development, Slough, UK.
Ian Whitehall is Sales Director for SP Industries, based at Genevac in Ipswich. Ian is recognised worldwide as anindustry expert in laboratory automation.
