In the previous article, the design of a freeze-drying cycle was described based on product behavior and thermal limits. That cycle is carried out through two drying stages — primary and secondary drying — which remove free and bound water from the product.
Primary Drying (& How It Removes Freeze Ice)
The drying portion of freeze-drying is actually a two-part process consisting of Primary Drying and Secondary Drying. The bulk of water removed from the product during freeze-drying is via sublimation of all of the free ice crystals during the primary drying step. Organic solvents are also removed during primary drying.
Primary drying (sublimation) is a slow process conducted at cooler temperatures, safely below the product’s critical collapse temperature. Sublimation requires heat energy to drive the phase change process from solid to gas. All three methods of heat transfer – conduction, convection and radiation, must be considered when freeze-drying a product.
In a simple manifold dryer, heat is transferred to the flask/product primarily through convection and radiation from the surrounding environment. With little control over heat flow into the product, it is more difficult to control the process. When working with products with low collapse temperatures, it may be necessary to wrap or insulate the flask to slow down the rate of heat transfer and avoid collapse.
In a shelf freeze dryer, it is important to maximize the surface contact of the product/container/tray with the shelf for good conductive heat transfer. However, the effects of radiation and convection also need to be considered for product uniformity and process control purposes.
Radiant heat from the inside walls of the product chamber will cause product/vials on the perimeter of the shelf to dry more quickly than product in the center of the shelf (known in freeze-drying as the “edge effect”). Radiation coming through the acrylic doors commonly used on pilot and R&D freeze dryers has an even greater effect and product located in the front of these dryers will typically dry the fastest of all. For this reason, production freeze dryers are designed with metal doors and small view ports. A piece of aluminum foil can be hung in front of the product on the inside of a pilot freeze dryer as a shield – of course this will block the view of the product and not allow observation during the process.
Because shelf contact is often inconsistent, convective heat transfer can help promote uniform product drying. System pressures in the 100 mTorr to 300 mTorrrange will usually promote an adequate amount of convection. At very lowsystem pressures less than 50 mTorr, there are fewer gas molecules present to provide convection and uneven / slower drying is likely and maintaining vacuum control can become an issue.
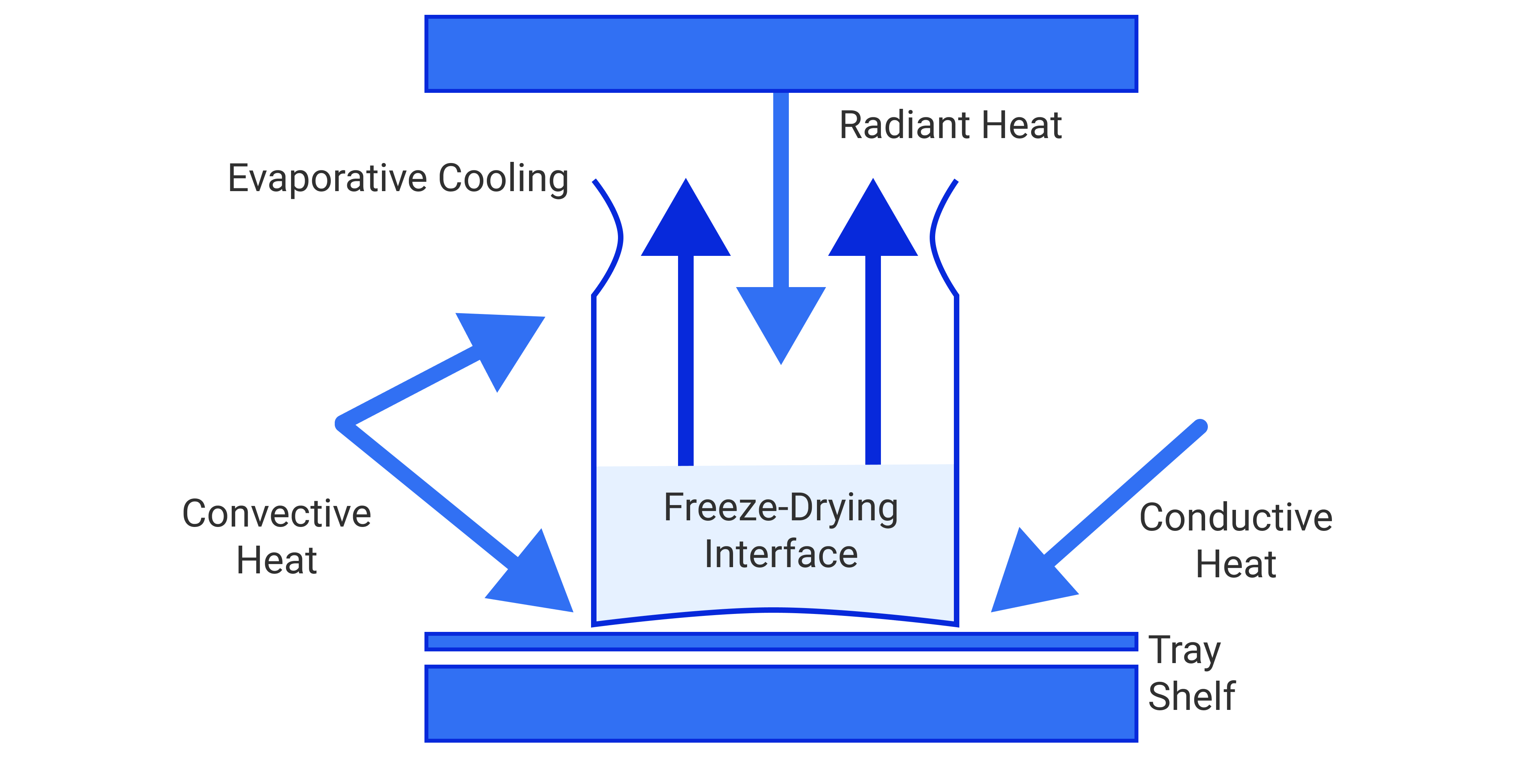
Primary drying is a top-down process with a well-defined sublimation front moving through the product as it dries. Above the ice surface interface is dried product, or “cake”; below the interface is product with ice crystals still remainingto be sublimed. At the end of primary drying when all of the free ice crystals have been sublimed, the product will appear to be dried. However, the moisture content can still be in the 5-10% range due to the presence of “sorbed” water molecules attached to the product.
Pressure & Temperature During Primary Drying
As mentioned earlier, each frozen product has a unique critical temperature. It is necessary to keep the product temperature safely below this critical temperature during primary drying to avoid collapse. The product temperature is dependent on the vapor pressure at the ice interface and in turn, this vapor pressure is dependent on both the rate of heat transfer into the product (which is controlled by adjusting the shelf temperature) and the system vacuum level set point.
Once a target product temperature is identified (typically several degrees safely colder than the critical temperature), the only two variables left to determine/control are the shelf temperature and system vacuum level. During primary drying, the system pressure and the shelf temperature are set and controlled in combination to yield the appropriate product temperature.
A recommended approach is to first set the system pressure using the vapor pressure over ice table (below). The product temperature is monitored using thermocouples and then the shelf temperature set point is slowly increased until the product reaches its target temperature. When the target product temperature is obtained, the shelf temperature is held constant for the balance of primary drying. Certain products with high resistance to vapor flow in the dried portion of the cake may require that the shelf temperature be reduced towards the end of primary drying to keep the product temperature at its target and to avoid collapse.
It is not recommended to increase the shelf temperature arbitrarily and repetitively during primary drying, as is seen on some older legacy cycles.
Using the vapor pressure of ice table is a scientific way to determine an appropriate pressure for freeze-drying. A general guideline is to choose a system pressure that is 20% to 30% of the vapor pressure of ice at the target product temperature. When the vacuum level set point is deeper than the vapor pressure of ice at the current product temperature, sublimation can take place. Typically, vacuum levels for freeze-drying are between 50mTorr and 300mTorr with 100mTorr to 200mTorr being the most common range.
With the temperature and pressure parameters set, primary drying is then continued for a length of time sufficient for all of the ice crystals to be sublimed.
As can be seen in the table below, temperature and pressure have a direct relationship, the lower the temperature of the ice, the lower the saturated vapor pressure over it.
Because most commercial freeze dryers cannot consistently control vacuum much below 30mTorr, at very cold product temperatures (less than -40 °C), it becomes impossible to have a system pressure set point that is 20% to 30 % of the vapor pressure of ice. Freeze-drying occurs extremely slowly at these cold product temperatures and process times may extend for 5 to 7 days or longer.
With manifold freeze-drying, the process is driven by the system pressure set point and the ambient temperature in the room. Because of the lack of control over the rate of heat transfer into the product, most manifold dryers are operated conservatively at lower pressures to help keep the product temperature lower.
| Temp | Vapor Pressure | ||
|---|---|---|---|
| [˚C] | [milliTorr] | [milliBar] | [pascal] |
| 0 | 4,584.000 | 6.1115 | 611.148 |
| -2 | 3,883.000 | 5.1769 | 517.689 |
| -4 | 3,281.000 | 4.3743 | 437.429 |
| -6 | 2,765.000 | 3.6864 | 368.635 |
| -8 | 2,325.000 | 3.0997 | 309.974 |
| -10 | 1,949.000 | 2.5984 | 259.845 |
| -12 | 1,630.000 | 2.1731 | 217.315 |
| -14 | 1,359.000 | 1.8118 | 181.185 |
| -16 | 1,130.000 | 1.5065 | 150.654 |
| -18 | 936.800 | 1.2490 | 124.896 |
| -20 | 774.400 | 1.0324 | 103.245 |
| -22 | 638.200 | 0.8509 | 85.086 |
| -24 | 524.300 | 0.6990 | 69.901 |
| -26 | 429.400 | 0.5725 | 57.248 |
| -28 | 350.500 | 0.4673 | 46.729 |
| -30 | 285.100 | 0.3801 | 38.010 |
| -32 | 231.200 | 0.3082 | 30.824 |
| -34 | 186.800 | 0.2490 | 24.905 |
| -36 | 150.300 | 0.2004 | 20.038 |
| -38 | 120.600 | 0.1608 | 16.079 |
| -40 | 96.300 | 0.1284 | 12.839 |
| -42 | 76.700 | 0.1023 | 10.226 |
| -44 | 60.800 | 0.0811 | 8.106 |
| -46 | 48.000 | 0.0640 | 6.399 |
| -48 | 37.700 | 0.0503 | 5.026 |
| -50 | 29.500 | 0.0393 | 3.933 |
| -52 | 23.000 | 0.0307 | 3.066 |
| -54 | 17.900 | 0.0239 | 2.386 |
| -56 | 13.800 | 0.0184 | 1.840 |
| -58 | 10.600 | 0.0141 | 1.413 |
| -60 | 8.100 | 0.0108 | 1.080 |
| -62 | 6.160 | 0.0082 | 0.821 |
| -64 | 4.660 | 0.0062 | 0.621 |
| -66 | 3.510 | 0.0047 | 0.468 |
| -68 | 2.630 | 0.0035 | 0.351 |
| -70 | 1.960 | 0.0026 | 0.261 |
| -72 | 1.450 | 0.0019 | 0.193 |
| -74 | 1.060 | 0.0014 | 0.141 |
| -76 | 0.780 | 0.0010 | 0.104 |
| -78 | 0.570 | 0.0008 | 0.076 |
| -80 | 0.410 | 0.0005 | 0.055 |
| -82 | 0.290 | 0.0004 | 0.039 |
| -84 | 0.210 | 0.0003 | 0.028 |
| -86 | 0.150 | 0.0002 | 0.020 |
| -88 | 0.100 | 0.0001 | 0.013 |
| -90 | 0.072 | 0.0001 | 0.010 |
Determination of the End of Primary Drying
Several analytical methods are available for determining that primary drying is complete. The most basic method is to monitor the product temperature with a thermocouple probe. The measured product temperature will be colder than the shelf temperature set point during active primary drying because the heat from the shelf is being used for the sublimation phase change. When sublimation of ice crystals is complete, the product temperature will increase and approach the shelf temperature. When the product temperature equals the shelf temperature, it can be inferred that primary drying is complete.
Note: the specific vial that contains the thermocouple wire will typically dry faster than the other vials on the shelf because the wire will conduct more heat into that specific vial. Similarly, if bulk drying, the area around the thermocouple wire will dry more quickly than other areas in the product tray. It is important to allow a modest amount of additional drying time (30 min to 2 hrs, depending on the product characteristics) after the product thermocouple temperature increases to ensure that all of the ice in the entire batch of product has been completely sublimated.
Because product will dry from the top down, the tip of the thermocouple should always be placed at the very bottom and center of the container. It is OK if the thermocouple touches the bottom of the container. If drying in vials, it is good practice to insert the thermocouple in a vial located in the middle of the shelf. Radiant heating effects will cause vials/product on the perimeter of the shelf to dry more quickly.

Additional primary drying endpoint determination tools are available on larger freeze dryers equipped with advanced process control systems. One such method entails comparison of parallel pressure readings between a Pirani gauge and a capacitance manometer. A capacitance manometer always gives a true pressure reading in the product chamber. The Pirani gauge, however, will give a false high reading in the presence of water vapor. When the Pirani pressure reading decreases and approaches the true pressure reading of the capacitance manometer, little or no water vapor is present, and it can be concluded that primary drying is complete.
Another tool is available with freeze dryer designs that have external condensers. An isolation valve can be added to the vapor port that connects the product chamber to the condenser. This valve can be closed for a short period of time and the subsequent rise in pressure in the product chamber can be measured. When this pressure rise approaches zero, no more water vapor is being generated via sublimation.
LyoFlux® TDLAS (Tunable Diode Laser Absorption Spectroscopy) is an advanced tool which measures the amount of water vapor flowing through the vapor port between the product chamber and condenser.
Secondary Drying (& Removing Bound Moisture)
In addition to the free ice that is sublimed during primary drying, there remains a substantial amount of water molecules that are bound to the product. This is the water that is removed (desorbed) during secondary drying. Since all of the free ice has been removed in primary drying, the product temperature can now be increased considerably without fear of melting or collapse.
Secondary drying actually starts during the primary phase, but at elevated temperatures (typically in the 30 °C to 50 °C range), desorption proceeds much more quickly. Secondary drying rates are dependent on the product temperature. System vacuum may be continued at the same level used during primary drying; lower vacuum levels will not improve secondary drying times.
Amorphous products may require that the temperature increase from primary to secondary drying be controlled at a slow ramp rate to avoid collapse.
Secondary drying is continued until the product has acceptable moisture content for long term storage. Depending on the application, moisture content in fully dried products is typically between 0.5% and 3%. In most cases, the drier the product, the longer its shelf life will be. However, certain complex biological products may actually become too dry for optimum storage results and the secondary drying process should be controlled accordingly.
During secondary drying, a “sample thief” mechanism may be used to periodically remove vials from the freeze dryer for residual moisture content determination.

Once both free ice and bound moisture have been removed, the freeze-drying process is complete.
In the next article, the focus will shift to optimizing the cycle, scaling it to production, and maintaining product stability during storage.










